| Synonyms |
Click to Show/Hide the Synonyms of This DIG
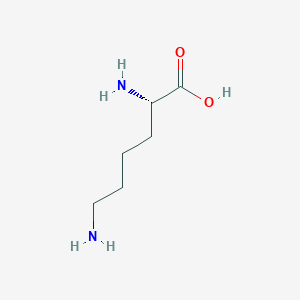
L-lysine; lysine; 56-87-1; h-Lys-oh; lysine acid; (S)-Lysine; (2S)-2,6-diaminohexanoic acid; Aminutrin; L-(+)-Lysine; alpha-Lysine; (S)-2,6-Diaminohexanoic acid; Hydrolysin; (S)-2,6-Diaminocaproic acid; Lysinum [Latin]; L-lys; Lisina [Spanish]; L-Norleucine, 6-amino-; Lysine, L-; Lysinum; (S)-alpha,epsilon-Diaminocaproic acid; Lysine [USAN:INN]; LYS (IUPAC abbreviation); L-2,6-Diaminocaproic acid; lysin; Hexanoic acid, 2,6-diamino-, (S)-; lys; a-Lysine; 2,6-Diaminohexanoic acid, (S)-; L-Lysin; BRN 1722531; CHEBI:18019; Poly-L-lysine; AI3-26523; 6-ammonio-L-norleucine; 25104-18-1; 12798-06-0; L-Lysine base; MFCD00064433; HSDB 2108; L-Lysine, 98%; L-2,6-Diaminocaproate; Gidrolizin; Hydrolysine; Lisina; L-LYSINE, MONOACETATE; (+)-S-Lysine; 3H-Lysine; L-Lysine, homopolymer; Lysine homopolymer, L-; 2,6-diaminohexanoate; (S)-(+)-Lysine; EINECS 200-294-2; lysina; L-Lysine, labeled with tritium; Ketporofen lysine; L-aLysine; .alpha.-Lysine; 1ozv; 1yxd; 3h-l-lysine; 6-amino-Aminutrin; NCGC00164527-01; Poly-(L-lysine); H-Lys; (-)-lysine; 6-amino-L-Norleucine; PubChem23203; a,e-Diaminocaproic acid; Lysine (USAN/INN); Alpha-poly-(L-lysine); L-2,6-Diainohexanoate; D-2,6-Diaminohexanoate; (S)-a,e-Diaminocaproate; L-103; DSSTox_CID_3232; L-Lysine, >=97%; bmse000043; bmse000914; Epitope ID:136017; (S)-2,6-Diaminohexanoate; L-2,6-Diainohexanoic acid; CHEMBL8085; DSSTox_RID_76935; DSSTox_GSID_23232; GTPL724; (S)-2,6-diamino-Hexanoate; (S)-a,e-Diaminocaproic acid; 26714-32-9; 4-04-00-02717 (Beilstein Handbook Reference); L-Lysine, analytical standard; L-Lysine, >=98%, FG; DTXSID6023232; (S)-2,6-diamino-Hexanoic acid; L-Lysine, >=98% (TLC); BDBM217367; (2S)-2,6-Diamino-hexanoic acid; ACT02654; HY-N0469; L-H2N(CH2)4CH(NH2)COOH; ZINC1532522; Tox21_112158; ANW-32594; Ethyl3,5-dichloro-4-propoxybenzoate; s5630; .alpha.,.epsilon.-Diaminocaproic acid; AKOS006239081; AKOS015855172; CCG-266180; CS-W019758; DB00123; EBD2198138; MCULE-3226089160; SB23228; CAS-56-87-1; L-Lysine solution, purum, 50% in H2O; NCGC00166296-02; 20166-34-1; AC-14492; AS-11733; (S)-.alpha.,.epsilon.-Diaminocaproic acid; AB0010643; L-Lysine, crystallized, >=98.0% (NT); AM20100376; L0129; L-Lysine, Vetec(TM) reagent grade, >=98%; A20652; C00047; D02304; M-6169; 59617-EP2272516A2; 59617-EP2281821A1; 59617-EP2295436A1; 59617-EP2298762A2; 59617-EP2298773A1; 59617-EP2301919A1; 59617-EP2308828A2; 59617-EP2308852A1; 064L433; J-521651; (S)-2,6-Diaminocaproic acid;(S)-(+)-Lysine;Lysine; Q20816880; F0001-1472; UNII-0O72R8RF8A component KDXKERNSBIXSRK-YFKPBYRVSA-N; UNII-AI4RT59273 component KDXKERNSBIXSRK-YFKPBYRVSA-N; UNII-K3Z4F929H6 component KDXKERNSBIXSRK-YFKPBYRVSA-N; UNII-N7U7BXP2OI component KDXKERNSBIXSRK-YFKPBYRVSA-N; 0013CD6B-1671-4369-B1BE-F531611E50C7
|
 click to show the detail info of this DBT
click to show the detail info of this DBT