Details of the Active Pharmaceutical Ingredient (API)
| General Information of API (ID: D00623) | |||||
|---|---|---|---|---|---|
| Name |
Simeprevir
|
||||
| Synonyms |
Click to Show/Hide the Synonyms of This API
SIMEPREVIR; Olysio; 923604-59-5; TMC435350; TMC-435; TMC 435; TMC 435350; TMC435; TMC-435350; CHEMBL501849; UNII-9WS5RD66HZ; (1R,4R,6S,7Z,15R,17R)-N-cyclopropylsulfonyl-17-[7-methoxy-8-methyl-2-(4-propan-2-yl-1,3-thiazol-2-yl)quinolin-4-yl]oxy-13-methyl-2,14-dioxo-3,13-diazatricyclo[13.3.0.04,6]octadec-7-ene-4-carboxamide; (2R,3aR,10Z,11aS,12aR,14aR)-N-(cyclopropylsulfonyl)-2-({7-methoxy-8-methyl-2-[4-(1-methylethyl)-1,3-thiazol-2-yl]quinolin-4-yl}oxy)-5-methyl-4,14-dioxo-2,3,3a,4,5,6,7,8,9,11a,12,13,14,14a-tetradecahydrocyclopenta[c]cyclopropa[g][1,6]diazacyclotetradecine-12a(1H)-carboxamide; 9WS5RD66HZ; HCV-PI; SCHEMBL826061; GTPL7367; C38H47N5O7S2; CHEBI:134743; N-cyclopropylsulfonyl-[[2-(4-isopropylthiazol-2-yl)-7-methoxy-8-methyl-4-quinolyl]oxy]-methyl-dioxo-[?]carboxamide; 4041AH; BDBM50336504; ZINC85540268; AKOS025401966; CCG-270435; AC-27651; AS-56205; TMC-00435350; TMC-435, TMC-435350; W-6083; US8741926, 47; US8754106, 47; Q7517689; (2R,3aR,10Z,11aS,12aR,14aR)-N-(Cyclopropylsulfonyl)-2-({7-methoxy-8-methyl-2-[4-(propan-2-yl)-1,3-thiazol-2-yl]quinolin-4-yl}oxy)-5-methyl-4,14-dioxo-2,3,3a,4,5,6,7,8,9,11a,12,13,14,14a-tetradecahydrocyclopenta[c]cyclopropa[g][1,6]diazacyclotetradecine-12a(1H)-carboxamide; (2R,3aR,10Z,12S,13R,15aR)-N-(Cyclopropylsulfonyl)-2-[[2-(4-isopropylthiazole-2-yl)-7-methoxy-8-methyl-4-quinolyl]oxy]-5-methyl-4,15-dioxo-2,3,3a,4,5,6,7,8,9,12,13,14,15,15a-tetradecahydro-12,13-methano-5,14-diaza-1H-cyclopentacyclotetradecene-13-carboxamide; (2R,3aR,11aS,12aR,14aR)-N-(cyclopropylsulfonyl)-2-(2-(4-isopropylthiazol-2-yl)-7-methoxy-8-methylquinolin-4-yloxy)-5-methyl-4,14-dioxo-1,2,3,3a,4,5,6,7,8,9,11a,12,12a,13,14,14a-hexadecahydrocyclopenta[c]cyclopropa[g][1,6]diazacyclotetradecine-12a-carboxamide; (2R,3aR,11aS,12aR,14aR,Z)-N-(cyclopropylsulfonyl)-2-((2-(4-isopropylthiazol-2-yl)-7-methoxy-8-methylquinolin-4-yl)oxy)-5-methyl-4,14-dioxo-1,2,3,3a,4,5,6,7,8,9,11a,12,12a,13,14,14a-hexadecahydrocyclopenta[c]cyclopropa[g][1,6]diazacyclotetradecine-12a-carboxamide; (2R,3aR,11aS,12aR,14aR,Z)-N-(cyclopropylsulfonyl)-2-(2-(4-isopropylthiazol-2-yl)-7-methoxy-8-methylquinolin-4-yloxy)-5-methyl-4,14-dioxo-1,2,3,3a,4,5,6,7,8,9,11a,12,12a,13,14,14a-hexadecahydrocyclopenta[c]cyclopropa[g][1,6]diazacyclotetradecine-12a-carboxamide; N-[17-[2-(4-isopropylthiazole-2-yl)-7-methoxy-8-methylquinolin-4-yloxy]-13-methyl-2,14-dioxo-3,13-diazatricyclo [13.3.0.0''4,6]octadec-7-ene-4-carbonyl](cyclopropyl)sulfonamide
|
||||
| Clinical Status |
Approved
|
||||
| Disease Indication | Viral hepatitis | ICD-11: 1E51 | [1] | ||
| PubChem CID | |||||
| Formula |
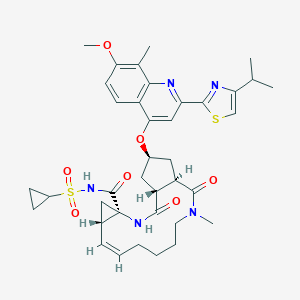
C38H47N5O7S2
|
||||
| Canonical SMILES |
CC1=C(C=CC2=C1N=C(C=C2O[C@@H]3C[C@@H]4[C@@H](C3)C(=O)N(CCCC/C=C\\[C@@H]5C[C@]5(NC4=O)C(=O)NS(=O)(=O)C6CC6)C)C7=NC(=CS7)C(C)C)OC
|
||||
| InChI |
1S/C38H47N5O7S2/c1-21(2)30-20-51-35(40-30)29-18-32(26-13-14-31(49-5)22(3)33(26)39-29)50-24-16-27-28(17-24)36(45)43(4)15-9-7-6-8-10-23-19-38(23,41-34(27)44)37(46)42-52(47,48)25-11-12-25/h8,10,13-14,18,20-21,23-25,27-28H,6-7,9,11-12,15-17,19H2,1-5H3,(H,41,44)(H,42,46)/b10-8-/t23-,24-,27-,28-,38-/m1/s1
|
||||
| InChIKey |
JTZZSQYMACOLNN-VDWJNHBNSA-N
|
||||
| Click to Show/Hide the Molecular Data (Structure/Property) of This API | |||||
| Structure |
<iframe style="width: 300px; height: 300px;" frameborder="0" src="https://embed.molview.org/v1/?mode=balls&cid=24873435"></iframe>
|
 |
|||
| 3D MOL is unavailable | 2D MOL | ||||
| Physicochemical Properties | Molecular Weight | 749.9 | Topological Polar Surface Area | 194 | |
| XlogP | 4.8 | Complexity | 1490 | ||
| Heavy Atom Count | 52 | Rotatable Bond Count | 8 | ||
| Hydrogen Bond Donor Count | 2 | Hydrogen Bond Acceptor Count | 10 | ||
| Full List of Drug Formulations (DFMs) Containing This API | ||||||
|---|---|---|---|---|---|---|
| Simeprevir 150 mg capsule | Click to Show/Hide the Full List of Formulation(s): 1 Formulation(s) | |||||
| Drug Formulation 1 |
DFM Info
 click to show the detail info of this DFM click to show the detail info of this DFM
|
|||||
| All DIGs | Click to Show/Hide the Full List of DIGs in This DFM
Lactose monohydrate; Sodium lauryl sulfate; Magnesium stearate; Ferrosoferric oxide; Titanium dioxide; Croscarmellose sodium; Silicon dioxide; Gelatin, unspecified; Shellac
|
|||||
| Dosage Form | Oral Capsule | |||||
| Company | Janssen Products | |||||
| DIG(s) with Biological Activity | ||||||
| DIG Name | DIG Info | Representative Biological Activity of This DIG | REF | |||
| Sodium lauryl sulfate | DIG Info | Solute carrier SLCO2B1 (Ki = 1.98 uM) | [2] | |||
| Magnesium stearate | DIG Info | Albendazole monooxygenase (Protein expression downregulation) | [3] | |||
| Carmellose sodium | DIG Info | Albendazole monooxygenase (Protein expression upregulation) | [3] | |||
| Silicon dioxide | DIG Info | Albendazole monooxygenase (Protein expression downregulation) | [3] | |||
| Simeprevir Sodium eq 150mg base capsule | Click to Show/Hide the Full List of Formulation(s): 1 Formulation(s) | |||||
| Drug Formulation 1 |
DFM Info
 click to show the detail info of this DFM click to show the detail info of this DFM
|
|||||
| All DIGs | Click to Show/Hide the Full List of DIGs in This DFM
Colloidal Anhydrous Silica; Croscarmellose Sodium; Lactose Monohydrate; Magnesium Stearate; Sodium Lauryl Sulphate; Gelatin; Titanium Dioxide (E171); Iron Oxide Black (E172); Shellac (E904)
|
|||||
| Dosage Form | Capsule | |||||
| Company | Janssen Prods | |||||
| DIG(s) with Biological Activity | ||||||
| DIG Name | DIG Info | Representative Biological Activity of This DIG | REF | |||
| Sodium lauryl sulfate | DIG Info | Solute carrier SLCO2B1 (Ki = 1.98 uM) | [2] | |||
| Gelatin | DIG Info | Mephenytoin 4-hydroxylase (EC50 = 20.5 uM) | [4] | |||
| Magnesium stearate | DIG Info | Albendazole monooxygenase (Protein expression downregulation) | [3] | |||
| Carmellose sodium | DIG Info | Albendazole monooxygenase (Protein expression upregulation) | [3] | |||
If you find any error in data or bug in web service, please kindly report it to Dr. Zhang and Dr. Mou.
