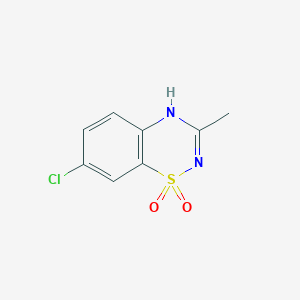
Details of the Active Pharmaceutical Ingredient (API)
| Full List of Drug Formulations (DFMs) Containing This API | ||||||
|---|---|---|---|---|---|---|
| Diazoxide 1.5g/30ml suspension | Click to Show/Hide the Full List of Formulation(s): 2 Formulation(s) | |||||
| Drug Formulation 1 |
DFM Info
 click to show the detail info of this DFM click to show the detail info of this DFM
|
|||||
| All DIGs | Click to Show/Hide the Full List of DIGs in This DFM
Sorbitol; Propylene Glycol; Carboxymethylcellulose Sodium; Sodium Benzoate; Methylparaben; Poloxamer 188; Propylparaben; Fd&C Red No. 40; Fd&C Yellow No. 6; Water; Alcohol; Hydrochloric Acid; Magnesium Aluminum Silicate
|
|||||
| Dosage Form | Suspension | |||||
| Company | Par Pharmaceutical | |||||
| DIG(s) with Biological Activity | ||||||
| DIG Name | DIG Info | Representative Biological Activity of This DIG | REF | |||
| Allura red AC dye | DIG Info | Solute carrier SLCO2B1 (Ki = 4.7 uM) | [1] | |||
| methylparaben | DIG Info | Carbonic anhydrase VII (Ki = 780 nM) | [2] | |||
| Sunset yellow FCF | DIG Info | Solute carrier SLCO2B1 (Ki = 68.4 uM) | [3] | |||
| Sodium benzoate | DIG Info | Carbonic anhydrase II (Ki = 30000 nM) | [4] | |||
| Allura red AC dye | DIG Info | Solute carrier SLCO2B1 (Ki = 2.59 uM) | [3] | |||
| Propylparaben sodium | DIG Info | Estrogen receptor alpha (EC50 = 38200 nM) | [5] | |||
| Hydrochloric acid | DIG Info | Carbonic anhydrase V (Ki = 156 nM) | [6] | |||
| Propylene glycol | DIG Info | Albendazole monooxygenase (Inhibition ratio < 20 %) | [7] | |||
| Carboxymethylcellulose sodium | DIG Info | Albendazole monooxygenase (EC50 = 12.6 uM) | [8] | |||
| Poloxamer 188 | DIG Info | Albendazole monooxygenase (IC50 = 60 uM) | [8] | |||
| Drug Formulation 2 |
DFM Info
 click to show the detail info of this DFM click to show the detail info of this DFM
|
|||||
| All DIGs | Click to Show/Hide the Full List of DIGs in This DFM
Sorbitol; Propylene Glycol; Magnesium Aluminum Silicate; Carboxymethylcellulose Sodium; Sodium Benzoate; Methylparaben; Poloxamer 188; Propylparaben; Alcohol; Hydrochloric Acid; Sodium Hydroxide
|
|||||
| Dosage Form | Suspension | |||||
| Company | Teva Pharmaceuticals Usa | |||||
| DIG(s) with Biological Activity | ||||||
| DIG Name | DIG Info | Representative Biological Activity of This DIG | REF | |||
| methylparaben | DIG Info | Carbonic anhydrase VII (Ki = 780 nM) | [2] | |||
| Sodium benzoate | DIG Info | Carbonic anhydrase II (Ki = 30000 nM) | [4] | |||
| Propylparaben sodium | DIG Info | Estrogen receptor alpha (EC50 = 38200 nM) | [5] | |||
| Hydrochloric acid | DIG Info | Carbonic anhydrase V (Ki = 156 nM) | [6] | |||
| Propylene glycol | DIG Info | Albendazole monooxygenase (Inhibition ratio < 20 %) | [7] | |||
| Carboxymethylcellulose sodium | DIG Info | Albendazole monooxygenase (EC50 = 12.6 uM) | [8] | |||
| Poloxamer 188 | DIG Info | Albendazole monooxygenase (IC50 = 60 uM) | [8] | |||
If you find any error in data or bug in web service, please kindly report it to Dr. Zhang and Dr. Mou.