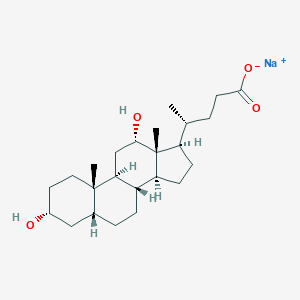
Details of the Drug Inactive Ingredient (DIG)
| Full List of Biological Targets of DIG (DBTs) Regulated by This DIG | ||||||
|---|---|---|---|---|---|---|
| G-protein coupled receptor (GPCR) | ||||||
| DBT Name: GPCR19 receptor (GPBAR1) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | EC50 = 4390 nM (estimated based on the structural similarity with CHEMBL3391714 ) | [1] | ||||
| Structural Similarity | Tanimoto coefficient = 1 | |||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | GPBAR_HUMAN | |||||
| Oxidoreductase (ORase) | ||||||
| DBT Name: Albendazole monooxygenase (CYP3A4) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 = 13 uM (tested by experiment) | [2] | ||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | CP3A4_HUMAN | |||||
| DBT Name: Cytochrome P450 2C9 (CYP2C9) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | EC50 = 118 uM (tested by experiment) | [2] | ||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | CP2C9_HUMAN | |||||
| DBT Name: Mephenytoin 4-hydroxylase (CYP2C19) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 = 359.7 uM (tested by experiment) | [2] | ||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | CP2CJ_HUMAN | |||||
| DBT Name: Debrisoquine 4-hydroxylase (CYP2D6) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 = 120.1 uM (tested by experiment) | [2] | ||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | CP2D6_HUMAN | |||||
| DBT Name: Cytochrome P450 3A5 (CYP3A5) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 = 224.7 uM (tested by experiment) | [2] | ||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | CP3A5_HUMAN | |||||
| Nuclear receptor (NR) | ||||||
| DBT Name: Farnesoid X-activated receptor (NR1H4) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | EC50 = 45000 nM (estimated based on the structural similarity with CHEMBL406393 ) | [3] | ||||
| Structural Similarity | Tanimoto coefficient = 1 | |||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | NR1H4_HUMAN | |||||
| DBT Name: Vitamin D3 receptor (VDR) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 > 50000 nM (estimated based on the structural similarity with CHEMBL406393 ) | [4] | ||||
| Structural Similarity | Tanimoto coefficient = 1 | |||||
| Tested Species | Homo sapiens (Human) | |||||
| UniProt ID | VDR_HUMAN | |||||
| Cell line (CELL) | ||||||
| DBT Name: Colon cancer Caco-2 cells (Caco-2) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 = 80300 nM (estimated based on the structural similarity with CHEMBL514674 ) | [5] | ||||
| Structural Similarity | Tanimoto coefficient = 1 | |||||
| Tested Species | Homo sapiens (Human) | |||||
| Cellosaurus ID | CVCL_0025 | |||||
| DBT Name: Hepatoblastoma HepG2 cells (HepG2) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 > 100000 nM (estimated based on the structural similarity with CHEMBL406393 ) | [6] | ||||
| Structural Similarity | Tanimoto coefficient = 1 | |||||
| Tested Species | Homo sapiens (Human) | |||||
| Cellosaurus ID | CVCL_0027 | |||||
| DBT Name: Duodenal cancer HuTu80 cells (HuTu80) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 = 82900 nM (estimated based on the structural similarity with CHEMBL406393 ) | [6] | ||||
| Structural Similarity | Tanimoto coefficient = 1 | |||||
| Tested Species | Homo sapiens (Human) | |||||
| Cellosaurus ID | CVCL_1301 | |||||
| DBT Name: Cervical cancer KB-3-1 cells (KB-3-1) | Click to Show/Hide | |||||
| Detailed Information |
DBT Info
 click to show the detail info of this DBT click to show the detail info of this DBT
|
|||||
| Experiment for Assessing the Biological Activity of the Studied DIG on This DBT | ||||||
| Biological Activity | IC50 > 100000 nM (estimated based on the structural similarity with CHEMBL406393 ) | [6] | ||||
| Structural Similarity | Tanimoto coefficient = 1 | |||||
| Tested Species | Homo sapiens (Human) | |||||
| Cellosaurus ID | CVCL_2088 | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Zhang and Dr. Mou.